(2) This is a two-dimensional image of something that is really three-dimensional some of those bonds would rise above the plane of the paper and some would project below it. Elements in the same column share properties so it should be no surprise that all of those elements form four bonds but only carbon will bond with another carbon because the atoms of those other elements are simply too large, even silicon, to link with another atom of the same element.Īll of the carbons and hydrogens are shown in this version of a structural formula, clearly showing how they relate to one another, but the image is misleading in two respects: (1) The carbons are not really linked in a straight line but rather in something that looks like a zig-zag line because of the orientation of the bonds of the carbon. The number and diversity of carbon compounds is so large that the study ofĬompounds in chemistry is divided into two great sub-disciplines: Organic Chemistry (the carbonĬompounds) and Inorganic Chemistry (everything else).Ĭarbon stands at the head of the 14th column of the Periodic Table, a column which also includes silicon, immediately below it, followed by germanium, tin, and lead. A carbon atom, however, is unique in that it can form bonds withĪs many as four other carbons, even forming double or triple bonds with another carbon, whichĬan link to yet more carbons to create a huge diversity of what can be incredibly complicatedĬarbon compounds. Oxygen will even form rather unstable (very reactive) triplets It’s easier than it looks.Some elements will form pairs of atoms of the same element, creating what are known as diatomic
#Naming compounds organic chemistry calculator how to
The best way to learn how to use the IUPAC system is to put it to work, not just memorize the rules. The rules enable us to not only name a compound from a given structure but also draw a structure from a given name. When these rules are followed, every unique compound receives its own exclusive name. The last alkyl group named is prefixed to the name of the parent alkane to form one word. For example, ethyl is listed before dimethyl the di- is simply ignored. These prefixes are not considered in determining the alphabetical order of the substituents.
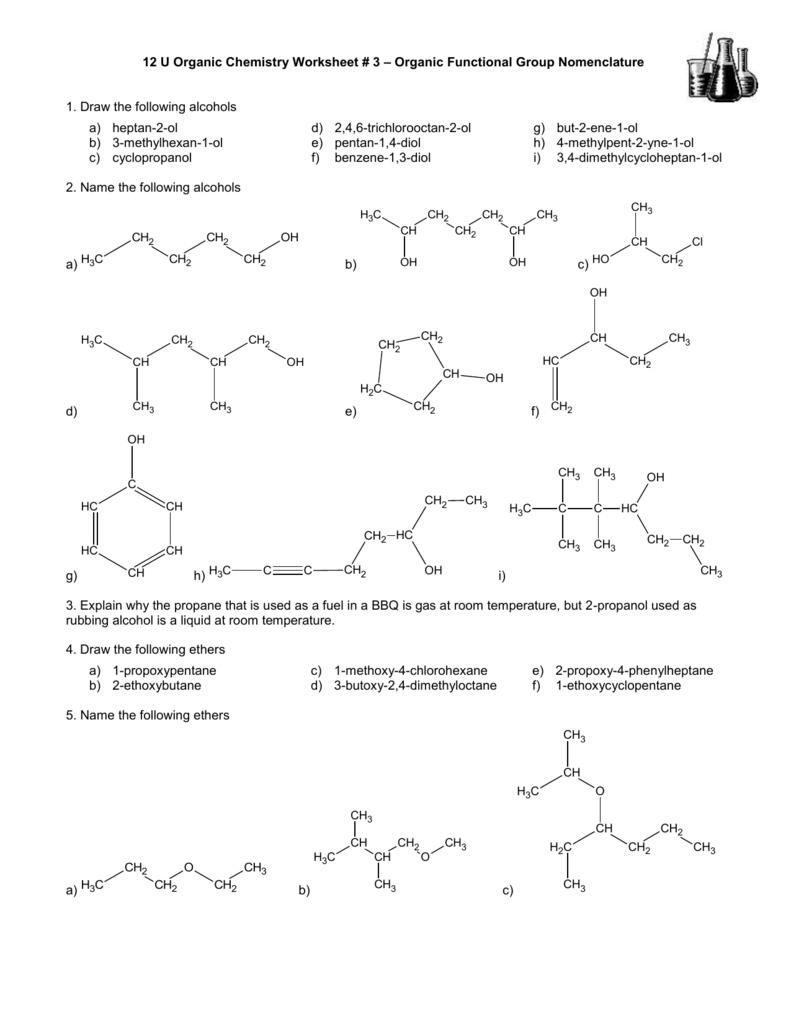
Moreover, the number of identical groups is indicated by the Greek prefixes di-, tri-, tetra-, and so on. If the same group appears more than once on the same carbon atom, the number of that carbon atom is repeated as many times as the group appears.

If the same alkyl group appears more than once, the numbers of all the carbon atoms to which it is attached are expressed. Place the names of the substituent groups in alphabetical order before the name of the parent compound. (The LCC need not be written in a straight line for example, the LCC in the following has five carbon atoms.)ģ. Hyphens are used to separate numbers from the names of substituents commas separate numbers from each other. Numbers are assigned in the direction that gives the lowest numbers to the carbon atoms with attached substituents. If the hydrocarbon is branched, number the carbon atoms of the LCC. This LCC, considered the parent chain, determines the base name, to which we add the suffix - ane to indicate that the molecule is an alkane.Ģ. Name alkanes according to the LCC (longest continuous chain) of carbon atoms in the molecule (rather than the total number of carbon atoms).

